Resource Centre
ASLM’s Resource Center is designed to help you keep up with the latest tool kits, regulations, guides and other information published by ASLM, partners, and global health regulatory bodies. Use the filters above to locate the information you need based on the ASLM project, resource type, or topic of interest. To find a specific resource, enter the resource name in the search bar. You may also use the search bar to enter key words related to the resources of your interest.
Through its Africa Task Force for Novel Coronavirus (AFTCOR) Laboratory Technical Working Group, the Africa Centres for Disease Control and Prevention (Africa CDC) has developed guidance on ‘Building Testing Capacity for Epidemic-Prone Diseases’. The aim of this guidance is to enhance the surveillance and diagnostic capacity of African Union (AU) member states. The guidance provides… Read More
Safe disposal of laboratory waste is a critical component of good laboratory practice. Molecular diagnostic test services, including HIV viral load (VL) and coronavirus disease 2019 (COVID-19) testing, have the potential to generate guanidinium thiocyanate (GTC), a potentially toxic compound, which requires specialised treatment and disposal. There are growing waste management challenges in many laboratories… Read More
In September 2021 the World Health Organization released updated guidance on maintaining essential health services during COVID-19, which provides direction on modifications and specific measures for safe delivery of HIV services, and considerations for transition towards restoration and recovery. Surveys of 101 countries have revealed substantial disruptions across all major health areas with up to… Read More
Essential diagnostic tests satisfy priority healthcare needs of a population and are selected based on disease prevalence, public health relevance, evidence of utility and comparative cost-effectiveness. Whereas essential testing services are such a critical piece in disease diagnosis, treatment monitoring, research and ongoing surveillance; pandemic containment measures, including repurposing testing capacity to the response, quarantine,… Read More
On 16 September 2021, ASLM’s LabCoP hosted another session in our series on manufacturers of COVID-19 diagnostics products. This session featured Siemens Healthineers and Thermo Fisher Scientific. The session focussed on innovations in SARS-CoV-2 to handle detection and surveillance of the fast-emerging variants of concern (VOC) and variants of interest (VOI) that have implications on… Read More
On 9 September 2021, this COVID-19 ECHO session was convened to discuss the evolving role of diagnostics in the COVID-19 pandemic response. Testing, when used effectively, is one critical tool that supports control of any disease outbreak including the COVID-19 pandemic. Swift case identification and contact tracing inevitably prevent further disease transmission. Researchers have worked… Read More
On 2 September 2021, this COVID-19 ECHO session was convened to share details on the COVIOS antigen rapid diagnostic test (Ag RDT) technology, status of regulatory approval, requirements/consumables to operate the test, and how Ag RDTs can be deployed to support testing programmes for COVID-19 epidemic control. COVIOS, developed by the Global Access Diagnostics, a… Read More
On 8 July 2021, this COVID-19 ECHO session was convened to discuss the perspective of Civil Society Organizations (CSOs) in leveraging on existing HIV community engagement to respond to COVID-19 challenges and opportunities. In this session, two CSOs, The National Forum for People living with HIV&AIDS Networks in Uganda (NAFOPHANO) and The Zimbabwe National Network… Read More
On 12 May 2021, this Extended ECHO session was convened to discuss the Global Fund COVID-19 funding mechanism, an opportunity for countries to request funds to strengthen their health systems in response while mitigating the effects of COVID-19 on other essential health services including HIV, tuberculosis (TB) and malaria. An innovative tool was discussed, that… Read More
On 13 May 2021, this COVID-19 ECHO session was convened to discuss a series of case studies on country experiences regarding the design and implementation of digital tools to manage the COVID-19 pandemic. The series was compiled by the Foundation for Innovative New Diagnostics (FIND) in collaboration with ministries of health and government partners. The goal was to promote… Read More
On 29 April, 2021 this COVID-19 ECHO session was convened to discuss tuberculosis (TB) and the COVID-19 pandemic. This panel discussion session was moderated by Rinuka Garde, Senior Advisor at the Clinton Health Access Initiative, and included three renowned experts in the field of TB and general lung health, including: Lucica Ditu, Executive Director Stop… Read More
On 15 April, 2021, ASLM’s LabCoP hosted the 13th session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring Abbott Rapid Diagnostics and Beckman Coulter Inc. In this session, Dr Anna Ruzhanskaya, Scientific Marketing Manager, CISEE, Beckman Coulter and Dr Kuku Appiah, Director Medical and Scientific Affairs-Abbot, share with participants some new developments… Read More
On 8 April, 2021 this COVID-19 ECHO session was convened to discuss the Africa CDC Trusted Travel Platform and how to create a ‘Trusted Network’ of COVID-19 testing laboratories in Africa. To support the reopening of boarders and economies without risking disease spread, Africa CDC and partners are championing the concept of Trusted Travel and… Read More
On 7 April, 2021, ASLM’s LabCoP hosted the 12th session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring Thermo Fischer Scientific. This session also focussed on mutation and variant surveillance of SARS-CoV-2. Without a robust, coordinated universal effort to identify and characterize emerging variants, societies risk suffering significant setbacks in health care… Read More
This presentation by the Clinton Health Access Initiative outlines the steps of an integration implementation framework for tuberculosis (TB) and HIV diagnostics, and related diseases, top tips and caveats (what could go wrong and lessons learned) from pilot countries on each step and area of implementation. It provides a deeper understanding of activities required for… Read More
This presentation by the Clinton Health Access Initiative outlines the need for point-of-care (POC) testing, how integrated testing improves access to POC, the benefits of integration, and considerations and implementation framework for integrated testing.
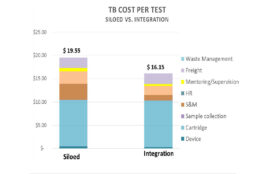
The GX Capacity Utilisation Analysis Tool is an Excel-based tool to assess the capacity utilisation of GeneXpert platforms for integrating tuberculosis (TB), HIV, HCV and HPV testing. It has been used to identify platforms running with full-capacity and with un-used capacity to inform integration decisions in Cameroon, Ethiopia, Indonesia, Malawi, Nigeria, Tanzania, Uganda, Zambia, and… Read More
The Integration Readiness Assessment Tool developed by Clinton Health Access Initiative and partners helps countries gather situation analysis information regarding an individual facility’s readiness to provide integrated testing for tuberculosis (TB), HIV and HPV. The assessment output of this MS Excel-based checklist allows for identification of sites where TB/HIV/HCV/HPV integration on the GeneXpert platform would… Read More
On 17 March, 2021 this COVID-19 ECHO session was convened to discuss outcomes, successes and lessons from implementing emergency SARS-CoV-2 external quality assessment proficiency testing in Africa. The African Joint taskforce on Coronavirus (AFTCOR) guidance objective 2 under Laboratory diagnosis is to ensure quality-assured testing for COVID-19 diagnosis, and strengthening reference laboratories and laboratory networks… Read More
On 2 March 2021 this COVID-19 ECHO session was convened to discuss the African experience in expanding COVID-19 testing using Antigen Rapid Diagnostic Tests (Ag RDT). It has been over one year since COVID-19 was declared a pandemic by WHO. There is still much concern that COVID-19 infections are increasing in some countries after several… Read More
On 12 February 2021, this COVID-19 ECHO session was convened for Prof Tulio de Oliveira to discuss the work of the Network of Genomic Surveillance of South Africa (NGS-SA) and the need to build a genomic network in Africa. From the beginning of the COVID-19 pandemic, the NGS-SA started genetic sequencing early enough by building… Read More
Laboratory diagnosis is an essential element of communicable disease surveillance, both for routine confirmation of infections and for the rapid identification of the cause of outbreaks and epidemics. An inaccurate diagnosis can have severe and expensive consequences such as inappropriate treatment or wastage of vaccines and test kits. Implementation of a strong quality management system… Read More
On 2 February, 2021 this COVID-19 ECHO session was convened to discuss expanding COVID-19 testing at schools, borders and at the workplace, in an effort to reopen economies. As many countries over the world are experiencing the second wave of COVID-19, forcing some to go back to stringent lockdown measures, countries are grappling with how… Read More
On 29 January 2021, ASLM’s LabCoP hosted the 10th session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring BD (US) and DiaSorin (Italy). Laboratory diagnosis remains the cornerstone for the control of COVID 19. New diagnostic methods with higher sensitivity and specificity, as well as faster results, are necessary and are continuously… Read More
On 22 January, 2021 this COVID-19 ECHO session was convened to discuss testing strategies for COVID-19 antigen rapid diagnostic tests (Ag RDT) and Africa CDC’s testing guidelines and recommendations. The start of 2021 has seen new challenges arise in the COVID-19 pandemic, with rapidly growing spread and new virus variants. To urgently address this, the… Read More
ASLM and Foundation for Innovative New Diagnostics (FIND) have produced a guide on how to develop and validate in-house RT-PCR tests for SARS-CoV-2. This guidance is essential, particularly in settings with limited diagnostics, including low- and middle-income countries (LMIC) where diagnostics are not immediately available during a pandemic. During the current COVID-19 pandemic the much… Read More
On 12 November 2020, this COVID-19 ECHO session was convened to discuss the launch of the SARS-CoV-2 antigen rapid test training package developed by ASLM and Africa CDC, in partnership with CHAI, AFENET, AMREF, and Last Mile Health. The training package, available to all African Union Member States, aims to support the scale-up of SARS-CoV-2… Read More
ASLM in collaboration with the Africa Centres for Disease Control and Prevention, and in partnership with the Clinton Health Access Initiative, Amref and Last Mile Health present the Quality Assurance Framework for SARS-CoV-2 Antigen Rapid Testing for Diagnosis of COVID-19. This framework aims to provide general technical guidance to African Union Members States on the… Read More
On 27 October 2020, this COVID-19 ECHO session was convened to discuss funding and procurement mechanisms for SARS-CoV-2 antigen rapid tests. As countries prepare to roll out SARS-CoV-2 antigen testing, understanding funding and procurement mechanisms will be critical in ensuring countries have access to adequate rapid tests. Several countries are planning to secure antigen tests… Read More
On 23 October, 2020 ASLM’s LabCoP hosted the ninth session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring STANDARD Q COVID-19 antigen rapid diagnostic test (Ag RDT). A new global partnership by WHO, The Global Fund, UNITAID, Africa CDC, FIND and other partners ensures that low-priced, high-quality Ag RDTs are available internationally… Read More
On 21 October, 2020 ASLM’s LabCoP hosted the ninth session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring Abbott’s Panbio Ag RDT, an in-vitro diagnostic test for the qualitative detection of SAR-CoV-2 antigen. In this session, Dr Kuku Appiah, Director, Medical & Scientific Affairs, Africa, Abbott Rapid Diagnostics discuss the Abbott Panbio… Read More
On 14 October, 2020 ASLM’s LabCoP dedicated a special session to digital solutions in support of health care providers, systems, and stakeholders, and how they have been applicable to the COVID-19 pandemic. It is increasingly clear that adopting digital technology and integrating it into Ministries of Health (MoH) policy is key for smooth coordination and… Read More
On 9 October 2020, this COVID-19 ECHO session and expert panel was convened to discuss what, why, and how the COVID-19 antigen rapid diagnostic tests (Ag RDTs) can be rolled out for optimal clinical and public health outcomes in Africa. Ag RDTs are fast and easy-to-deploy diagnostic methods that take 10 – 15 minutes to… Read More
On 5 November, 2020 this COVID-19 ECHO session was convened to discuss considerations of pooling specimens for testing by real-time RT-PCR. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) detection, also known as COVID-19 diagnosis, is performed using a molecular technique referred to as real-time reverse transcriptase-polymerase chain reaction (RT-PCR). This has remained the method of… Read More
As part of the global response to the COVID-19 pandemic, laboratory diagnosis has remained as the cornerstone to this intervention. Molecular assays performed on nasopharyngeal swabs or other upper respiratory tract specimens are the most commonly used and reliable tests for the diagnosis of COVID-19. A variety of RNA gene targets are used by different… Read More
Decentralising laboratory testing for COVID-19 to on a national scale will be effective if COVID-19 nucleic acid testing is extended to subnational levels. However, this expansion process from national to sub-national levels has to be balanced carefully so as to meet the increasing demanding for the COVID-19 testing service while maintaining a high quality of… Read More
On 18 September 2020, this COVID-19 ECHO session was convened to discuss antigen testing’s role in helping control the COVID-19 pandemic. Real time Polymerized Chain Reaction (rt-PCR) testing to detect SARS-CoV-2 has been the WHO recommended method to diagnose COVID-19. However, this method has not always been available at every tier of the laboratory network… Read More
On 17 September 2020, this COVID-19 ECHO session was convened to discuss self-collection of nasal specimens as a means to lowering the barrier to SARS-CoV-2 testing and access to care. Diagnostic testing for SARS-CoV-2 remains a challenge today, even months since the outbreak of the pandemic and with the emergence of several innovations. In the… Read More
On 13 August 2020, this COVID-19 ECHO session was convened to discuss optimization of diagnostic networks and their key role in responding to COVID-19. Optimised national diagnostic networks are key to increasing access to high-quality diagnostics. With the outbreak of the COVID-19 pandemic, some sub-Saharan African countries leveraged more than a decade of investments in… Read More
On 6 August 2020, this COVID-19 ECHO session was convened to discuss laboratory data management considerations for COVID-19 response. Long turnaround times (TAT) for laboratory test results and poor data quality are chronic problems for many testing services. Timely and accurate testing services for COVID-19 are essential for monitoring the spread of the pandemic and… Read More
On 15 July 2020, this COVID-19 ECHO session was convened to discuss the public health lab networks and SARS-CoV-2 testing challenges, strategies, and lessons learned from the USA. During emergencies like COVID-19 when laboratory testing services surge, there are risks of a health care system being overwhelmed. Well-structured strategies must be formulated to cope with… Read More
On 10 July 2020, ASLM’s LabCoP hosted the eighth session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring Roche Diagnostics International. Presentations were made by Dr Wim van der Helm, Head of Healthcare Development, Roche Diagnostics, Europe, Middle-East, Africa and Latin America, and Dr Matthias Strobl, Senior Clinical Science Leader, Infectious Diseases,… Read More
The World Health Organization has updated their interim operational guide for maintaining essential health services during an outbreak, to support countries preparedness and response during the COVID-19 pandemic. It supplements existing and future WHO guidance on the wider implications of COVID-19 for health systems, and cross-government strategies for responding to the COVID-19 outbreak. The updated… Read More
In this study, Robert Fischer, et al, present their assessment findings on N95 respirator decontamination and re-use for SARS-CoV-2 (24 April 2020), using four different decontamination methods – UV radiation (260 – 285 nm), 70º C dry heat, 70% ethanol and vaporized hydrogen peroxide (VHP). The authors recommend that N95 respirators be decontaminated and re-used… Read More
David Berlin, et al, present key clinical features of severe COVID-19 in the New England Journal of Medicine (21 May 2020). The authors start with a clinical description of a 50-year old COVID-19 patient. The authors recommendation is that the most important aspect of care is careful monitoring of respiratory status to determine whether endotracheal… Read More
The World Health Organization has published Version 2 (22 May 2020) of their guidance about HIV, viral hepatitis and STI programmes in relation to COVID-19. The guidance is intended to support national HIV and viral hepatitis programmes and their health workers, to provide guidance to people affected by HIV and viral hepatitis, and to maintain… Read More
The European Centre for Disease Prevention and Control (ECDC) has published this Rapid Risk Assessment Report on the association of paediatric inflammatory multisystem syndrome (PIMS) and SARS-CoV-2 infection in children (15 May 2020). ECDC concludes risk communication should emphasise that PIMS is a rare condition and that its potential link with COVID-19 is neither established… Read More
In Nature Communications, Chuyan Wang, et al (4 May, 2020) describe a human monoclonal antibody that neutralizes SARS-CoV-2 in cell culture. The authors highlight that the cross-neutralizing antibody targets a communal epitope on the virus and may offer potential for prevention and treatment of COVID-19.
LabCoP’s June ECHO session covered differentiated service delivery (DSD) for HIV care. DSD is a client-centered approach aimed at simplifying and adapting HIV services to different groups of people living with HIV, while reducing the burden on the health system. In recent months, the response to the COVID-19 pandemic has included adjustments to clinical care… Read More
On 18 June 2020, ASLM’s LabCoP hosted the sixth session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring PerkinElmer and Pro-Med. Stephanie Wilbraham, Market Development Manager, PerkinElmer, and Neil Barker, Co-Founder, Pro-Med Diagnostics, South Africa presented about the overview of their available SARS-CoV-2 laboratory test kit(s), performance, run time & other technical… Read More
On 11 June 2020, ASLM’s LabCoP hosted the fifth session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring KH Medical and Inqaba Biotec. Dr Aron Abera, Technical Support Manager, Inqaba Biotec, South Africa; Dr Julius Muhumuza, Technical Advisor, KH Medical Co., Ltd.; Mr Adam Hong, CEO, KH Medical Co., Ltd.; and Dr… Read More
The World Health Organization has published interim guidance on the use of masks in the context of COVID-19 for health settings and home care. This interim guidance provides detailed pragmatic advice to decision makers on the use of masks for healthy people in community settings based on risk, vulnerability, settings and feasibility.
In a retrospective study of hospitalized patients with COVID-19, by Joshua Barbosa et al, published in the New England Journal of Medicine, it shows that Hydroxychloroquine does not seem to have meaningful benefits on clinical outcome measures of mortality, lymphopenia, or neutrophil-to lymphocyte ratio improvement. The authors demonstrate that patients receiving Hydroxychloroquine appeared to have… Read More
On 2 June 2020, this COVID-19 ECHO session was convened to discuss establishing an integrated specimen referral system (SRS) for SARS-CoV-2 testing recommended to enable access to testing services across the entire health pyramid. Many countries struggled to operationalise this guidance and failed to create sustainable and efficient sample transportation systems. Due to various challenges,… Read More
In this MEDSCAPE commentary, Dr Paul Sax from Brigham and Women’s Hospital and Harvard Medical School summarises some of the most common questions regarding COVID-19 and HIV: Is COVID-19 a more severe disease in people with HIV? Do I need to come in for my regular blood tests and checkups? Do my antiretroviral medications protect… Read More
In this guideline, specifically prepared for COVID-19 pandemic, Africa CDC advises Member States to use the characteristics of the epidemic in their country to decide when and how to do contact tracing. Namely, in Epidemic Phase 1-2 where imported cases and cases in known transmission chains the guidance is to trace all contacts. In Epidemic… Read More
The Writing Group of the Johns Hopkins University and Johns Hopkins Hospital COVID-19 Treatment Guidance Working Group presents a compressive guidance for existing treatment for COVID-19. The guidance strongly recommends clinical trials for potential treatments before any use, how to use immune modulatory agents to treat COVID-19 such as IL-6R or IL-6 Monoclonal antibodies, intravenous… Read More
This study by Nathan Ford, et al. (20 March, 2020), published in the Journal of the International AIDS Society, presents the systematic review of clinical outcomes using antiretroviral drugs for the prevention and treatment of coronaviruses and planned clinical trials. Their review indicates that it is uncertain whether LPV/r and other antiretrovirals improve clinical outcomes… Read More
On 28 May, 2020 ASLM’s LabCoP hosted the fourth session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring Abbott. Presentations were made by Dr Claudio Galli, Associate Director, Medical Scientific Affairs Global, Abbott Core Laboratory, and Dr Liane Bauer, Director, Professional Services EMEA, Abbott Core Laboratory. They covered topics including detection of… Read More
During severe shortages of personal protective equipment (PPE) due to COVID-19, one solution to consider is decontamination and extended use of medical masks and N95/FFP2 respirators for health workers at the frontline. On 26 May, 2020 ASLM’s LabCoP hosted a session that covered these considerations and additional recommendations on homemade masks that have protection equal… Read More
On 20 May 2020, this COVID-19 ECHO session was convened to discuss procurement and the supply chain for COVID-19 diagnostics. Dr Lara Vojnov, Diagnostics Advisor at the World Health Organization, Geneva, and Dr Yenew Kebede, Head of Laboratory Services at Africa CDC presented about the need to coordinate and collaborate test availability and access; current… Read More
LabCoP’s May ECHO session covered the key considerations for expanding COVID-19 capacity to the sub-national level and featured the Ethiopian experience. During this session, participants learnt about the need to scale up diagnostic capacity; governance and coordination issues essential for expansion of COVID-19 testing; leveraging on existing capacity to cope with the needs; strategies for… Read More
The May 2020 Waste Management training session focused on SARS-CoV-2 laboratory biosafety guidance and featured the presentation of David Bressler, Health Scientist and Microbiologist at the International Laboratory Branch of the U.S. Centers for Disease Control and Prevention. Nearly all countries are scaling up COVID-19 testing as one of the main strategies to eliminate the… Read More
On 14 May 2020, this COVID-19 ECHO session was convened to assist the African laboratory community in designing and implementing an effective COVID-19 quality assured testing program. Mr Patrick Mateta, Vice President, Global Health Partnerships, at Clinical and Laboratory Standards Institute (CLSI) presented about how to plan and prepare for COVID-19 testing, the selection and… Read More
This article, published in STAT, lists the COVID-19 drugs and vaccines in development, to help inform efforts to treat or prevent COVID-19. Note: The article lists novel medicines, not repurposed medicines.
On 8 May 2020, ASLM’s LabCoP hosted part 2 of an ECHO session about COVID-19 serology tests. Serological diagnostic testing has been proposed to be useful in combination with molecular testing, in confirming COVID-19 infection or exposure to the virus. Serology tests also have the potential to be delivered in rapid format and at the… Read More
On 30 April 202, ASLM’s LabCoP hosted the third session in a series about manufacturers of COVID-19 molecular diagnostic tools, featuring Abbott and Bio-Rad. Dr Danijela Lucic, Senior Scientific Affairs Manager, Global Infectious Disease, Abbott Molecular, and Dr Marcus Neusser, EMEA Product Manager, Gene Expression, Bio-Rad, Germany, and Mr Richie Petronis, Product Manager, Bio-Rad, USA,… Read More
On 29 April 2020, ASLM’s LabCoP hosted this ECHO session on strategies to implement fast turn-around laboratory testing for control of COVID-19, presented by Dr Trevor Peter, Senior Director, Diagnostic Services, Clinton Health Access Initiative. Nucleic acid testing is the WHO-recommended method for detecting SARS-CoV-2. The global scale-up of COVID-19 diagnostics will depend on use… Read More
On 24 April 2020, Professor Rosanna Peeling and colleagues from the International Diagnostics Centre at the London School of Hygiene and Tropical Medicine pulled together evidence for and against the original set of recommendations for use of serologic tests. Prof Peeling was joined by Dr Yenew Kebede from Africa CDC and Dr Amadou Sall from… Read More
This paper describes the steps taken by Brazil before the first COVID-19 cases were identified in that country. The steps included creation of ordinances and laws, setting up of emergency operating centers, and clearly articulating how isolation and quarantine will be done. The paper also examines the advantages of these measures. Based on the Brazilian… Read More
This paper highlights important research questions regarding SARS-CoV-2 which include origin, virus transmission, viral shedding, diagnosis, treatment, vaccine development and viral pathogenesis. The paper proposes that research on SARS-CoV-2 and COVID-19, if it seeks to answer these important questions, will solidify the scientific basis on which important policy and scientific decisions are made to contain… Read More
This presentation by Prof Yazdan Yazdanpanah, published by the Global Research and Innovation Forum, presents a global roadmap for the control of COVID-19. The roadmap defines research priorities regarding how to stop the transmission of COVID-19, and how to mitigate its impact, as well as a governance framework from an ethics point of view. The… Read More
This study, by Xi He et al, published in Nature Medicine (15 April, 2020), reports that 44% of secondary COVID-19 cases were transmitted by people who had not yet developed symptoms. The findings emphasize that control measures should be adjusted to account for probable substantial presymptomatic transmission. Sensitive and specific molecular and serological testing is… Read More
Laboratories need to conduct verification or validation studies to confirm that new tests for COVID-19 diagnosis perform as intended. In this document co-authored by ASLM, FIND and the London School for Hygiene and Tropical Medicine, further key requirements for quality assurance are defined, including quality control and external quality assessment that laboratories should follow to… Read More
LabCoP’s April ECHO session featured presentations about maintaining HIV and TB testing in the context of COVID-19 from Dr George Alemnji, Senior Technical Advisor for Laboratory Services at the State Department Office of Global AIDS Coordinator (SGAC) and Health Diplomacy, US CDC; Dr Lara Vojnov, Diagnostics Advisor, WHO; and Dr Dennis Falzon, Medical Officer, World… Read More
This study by Dr Bin Cao, et al, 2020, in the New England Journal of Medicine describes a trial of lopinavir–ritonavir in adults hospitalised with severe COVID-19 where no benefit was observed with this treatment beyond standard care.
This systematic review of the efficacy and safety of antiretroviral drugs against SARS, MERS, or COVID-19 (initial assessment) was conducted by Dr Nathan Ford, et al, 2020. Their conclusion from the available evidence is that it is uncertain whether LPV/r and other antiretrovirals improve clinical outcomes or prevent infection among patients at high risk of… Read More
This report details the first full genome sequence of SARS-CoV-2 from Africa to be released, with genomic data generated and assembled, and phylogenetic analysis performed by ACEGID and NIMR scientists.
The Liverpool Drug Interactions Group (based at the University of Liverpool, UK), presents an exhaustive chart about common drug interactions with experimental COVID-19 therapies.
This March 2020 presentation of the WHO-China joint mission includes key findings on COVID-19. Epidemiologic and technical insights to contain the pandemic are also covered.
This modelling study by Marius Gilbert, et al, published in the Lancet, 2020, evaluates preparedness and vulnerability of African countries against importations of COVID-19. Their evaluation was based on WHO International Health Regulations (IHR) Monitoring and Evaluation (M&E) Framework, and on an indicator of vulnerability to emerging epidemics.
This article published in the New England Journal of Medicine by W. Guan, et al, 2020, describes the clinical characteristics of affected patients with COVID-19 in China. The findings show more frequent absence of fever in COVID-19 than in SARS-CoV (1%) and MERS-CoV infection (2%). This can have an implication if surveillance case definition focuses… Read More
This paper published in the New England Journal of Medicine by Neeltje van Doremalen, et al, 2020, describes the stability of viable SARS-CoV-2 on surfaces and in aerosols in comparison with SARS CoV-1. The results indicate that aerosol and fomite transmission of SARS-CoV-2 is plausible, as the virus can remain viable in aerosols for multiple… Read More
This article by Mark S. Lesney and published by MDedge, 2020, highlights the news that researchers have identified the structure of a spike protein that could turn out to be a potential vaccine target for the 2019-nCoV that causes COVID-19. The research further suggests that future antibody isolation and therapeutic design efforts will benefit from… Read More
On 16 April 2020, ASLM’s LabCoP hosted the second session in a series about manufacturers of COVID-19 molecular diagnostic tools. Dr Davide Manissero, Chief Medical Officer of Infection and Immune Diagnostics at QIAGEN, and Mr Riegardt Johnson, Field Application Scientist at Thermo Fisher Scientific presented about their available COVID-19 laboratory test kit(s), their status regarding… Read More
On 14 April 2020, ASLM’s LabCoP hosted the first session in a series about manufacturers of COVID-19 molecular diagnostics tools, including BD and Cepheid. Dr Charles Cooper, Global Vice President, Medical Affairs, Integrated Diagnostic Solutions at BD, and Ms Rujeko Tsomondo, Regional Application Lead at Cepheid presented about their available COVID-19 laboratory test kit(s), their… Read More
This report by WHO gives an overview on the use of licensed medicines for indications that have not been approved by regulatory authorities. It provides a scientific summary of key aspects to consider in off label use of medicines and the need to adhere to national regulations on the use of medicines. It also stipulates… Read More
This is an exhaustive PowerPoint presentation by Paolo Surico and Andrea Galeotti about the economics of a pandemic, using COVID-19 as a case study. The presentation sheds light on the science, health policies, economics and macroeconomic policies.
The European Centres for Disease Prevention and Control (ECDC) has published this technical report about safe handling of bodies of deceased persons with suspected or confirmed COVID-19 to be used by public health authorities in EU/EEA Member States and the United Kingdom. These considerations may help inform the approach in Africa.
This paper by Erin K. McCreary and Jason M. Pogue about COVID-19 treatment describes a review of early and emerging options. The authors summarize the current evidence as of 19 March 2020 to provide guidance on potential COVID-19 treatment options.
The Nigeria Centre for Disease Control published this self-isolation guidance for Nigerians on 18 March 2020 that providesdetailed description of what self-isolation means during COVID-19 pandemic and step-by-step instructions for adherence. This guidance may help inform the policies set forth my ministries of health of other African countries.
This paper by Chengzhi Yang & Zening Jin, 2020, indicated that individuals with underlying chronic cardiovascular diseases (CVDs) were both more susceptible to COVID-19 and more prone to critical conditions and death. Therefore, continued observations of the cardiovascular complications of the disease are needed.
On 8 April 2020, ASLM and LabCoP convened a special ECHO session on lessons the African continent can learn from the Korean experience in controlling COVID-19 Dr Seon Kui Lee, Director, Division of Risk Assessment & International Cooperation at the Korea Centers for Disease Control and Prevention (KCDC), shared an overview of the KCDC and… Read More
This paper by Shaobo Shi et al, 2020, explores the association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China.
Africa CDC has published this guidance and recommendations for community social distancing during the COVID-19 outbreak. Social distancing is an accepted strategy to delay and reduce the magnitude of outbreaks of pandemic influenza. It is necessary at the individual and community levels, because transmission occurs frequently from person-to-person.
This paper by David M. Morens and Anthony S. Fauci provides insight about the 1918 influenza pandemic and lessons learned, which could still apply in the 21st Century related to COVID-19, Ebola or other outbreaks.
This review about the epidemiology and pathogenesis of coronavirus disease (COVID-19) by Hussin A. Rothan et al, 2020, in the Journal of Autoimmunity highlights the symptoms, epidemiology, transmission, pathogenesis, phylogenetic analysis, and future directions to control the spread of this fatal disease.
This paper by Yixuan Wang et al, 2020 summarises the latest literature on genetic, epidemiological, and clinical features of COVID-19 compared to SARS and MERS, and emphasises special measures on diagnosis and potential interventions. This review can improve our understanding of the unique features of COVID-19 and enhance our control measures in the future.
WHO has published interim guidance for laboratory testing for coronavirus disease COVID-19 in suspected human cases. It also describes safety procedures for specimen collection and testing from suspected cases and contacts. Suspect cases should be screened for the virus with nucleic acid amplification tests (NAAT), such as RT-PCR. Download the guide and read more.
On 30 March 2020, ASLM and LabCoP convened a special ECHO session on the latest available technology to diagnose COVID-19, and recommendations for clinical care and surveillance. Dr Cassandra Kelly-Cirino, from the Foundation for Innovative New Diagnostics (FIND), and Prof Rosanna Peeling, from the London School of Hygiene & Tropical Medicine, gave an overview of… Read More
On 25 March 2020, ASLM and LabCoP convened an ECHO session on troubleshooting common challenges associated with establishing diagnostic testing for SARS-CoV-2 (the causative agent of COVID-19). Dr Jinal Bhiman, Director of the National Influenza Centre at the NICD, South Africa, presented practical solutions to address the most common challenges encountered by laboratory staff recently… Read More
WHO has published these recommendations for health workers and global health professionals who travel, to help them stay safe and minimise their chances of contracting the 2019-nCoV in the course of their work. Please download these documents below.