Sputum Pooling: Maximising Impact with an Old Tool
Tuberculosis (TB) has affected humanity for millennia. Observed on 24 March, World TB Day highlights both progress and remaining challenges in TB control. This year brings renewed optimism with new WHO guidelines recommending near point of care (NPOC) nucleic acid amplification tests (NAAT) and the use of tongue swabs and pooled sputum testing with existing molecular tests.
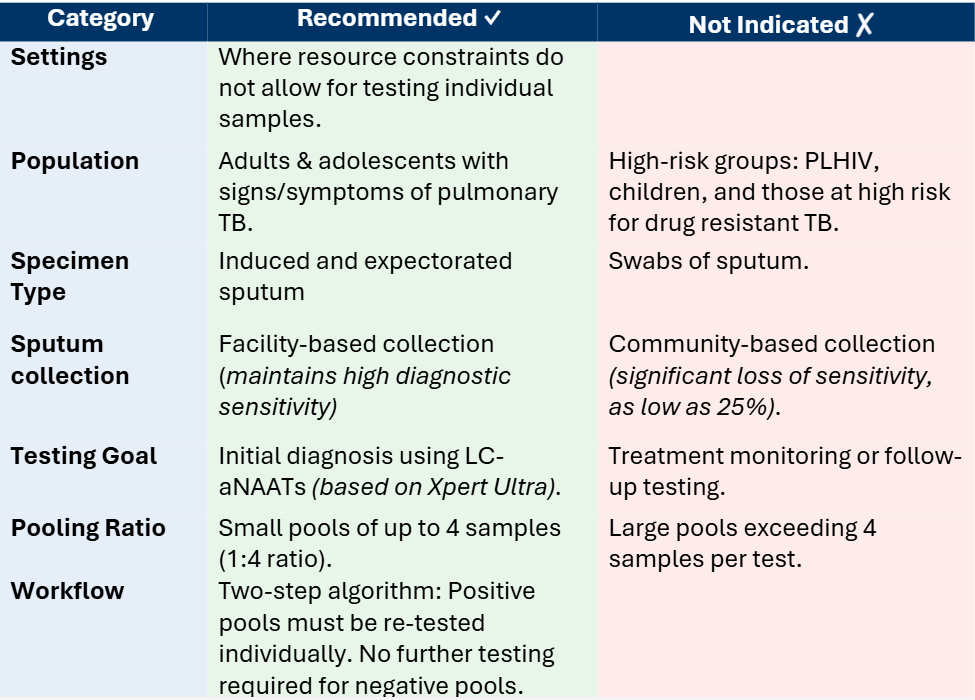
Specimen pooling, combining samples from several patients into one test, has been used for syphilis, HIV, COVID-19, blood donation screening, and influenza surveillance but is less common for TB due to concerns about lower diagnostic accuracy and higher false negatives. Recent studies showed that pooling four sputum samples for Xpert MTB/RIF Ultra testing resulted in 84.8% sensitivity and 98.1% specificity. Pooled testing lowered costs from $7.97 to $11.33 per individual test to $3.04 to $9.95. A minor drop in sensitivity (~3%) was deemed an acceptable trade-off to enable broader testing with low complexity automated nucleic acid amplification tests (LC-aNAAT) (Table 1).
Pooled sputum testing utilises the current LC-aNAAT infrastructure and personnel, resulting in minimal additional training requirements. Nevertheless, TB laboratories should assess the following factors to facilitate effective implementation and fully realise the cost benefits provided by the pooled sputum approach:
- Evaluations: Current recommendations are based on Xpert Ultra data. Further evaluations are needed for Truenat and TB-LAMP.
- Settings: Sputum pooling is not efficient in all contexts. Cost and time savings drop above 30% test positivity, with reviewed studies showing benefits up to 24%.
- Training: Staff familiar with LC-aNAAT require minimal retraining, but all staff should be instructed on maintaining a 1:4 pooling ratio, proper sample handling, quality control, pool assignment, and result interpretation.
- Workflows: Pooling adds steps to standard testing. Laboratories should refine workflows for sample splitting, labelling, and retesting, possibly piloting at select sites before broader implementation.
- Specimen quality: High quality samples are essential, therefore, training clinical staff and educating patients on producing a good quality sample is important.
- Result management: Update LIS to record both pool and individual IDs. Monitor turnaround times to quickly address workflow issues.
To support countries in implementing these new guidelines, WHO will soon release practical guidance and a toolkit outlining key steps to facilitate program uptake. ASLM has organised webinars to sensitise national programs and stakeholders on the performance characteristics of NPOC-NAAT and share early country experiences. Furthermore, ASLM has worked with 7 countries in Africa to support market shaping efforts, ensuring countries are prepared for procurement and deployment. ASLM will continue to provide targeted support to optimise national diagnostic networks, ensuring that NPOC-NAATs are placed where they will have the greatest impact. In addition, ASLM will provide technical assistance to refine workflow processes, strengthen quality assurance systems, and ensure high-quality testing for pooled sputa and swab-based testing on LC-aNAAT platforms.
As funding gaps challenge global health goals, sputum pooling is not just a technical work-around but a strategic necessity, enabling programs to “do more with less”. Optimising limited resources ensures that rapid TB detection is available to everyone, everywhere.

