In Q4 2025, the Laboratory Systems Strengthening Community of Practice (LabCoP) project, supported by the Gates Foundation and partner organizations, hosted four webinars. The sessions highlighted new diagnostic tools, launched key frameworks, and explored current developments in the dynamic funding landscape.
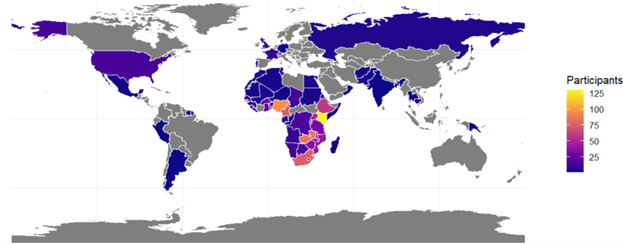
The webinars attracted 1,107 participants from 79 countries, averaging 276 attendees per session, including laboratory professionals, policymakers, and public health stakeholders. Median participation time was 56 minutes (IQR 31-76)
Fig 1. Heat Map Showing number of participants attending the 4 sessions by country
Session Highlights
Current external funding dynamics: impacts, implications and looking ahead
Two sessions focused on historical dependence on external funding, impacts of funding withdrawals, and current initiatives addressing short-term and long-term strategies. Mozambique shared progress in building resilient laboratory systems amid reduced funding; while Zambia discussed navigating the path forward via a forthcoming bilateral MOU with the United States Government.
These discussions highlighted the impact of funding freeze on almost all facets of general health and laboratory programs and hence the need for strategic domestic funding aligned with service needs to be taken as essential, rather than optional. External funding could be viewed as supplemental rather than core to protect programming.
Key considerations for transitioning to sustainable programs that countries should consider include stakeholder engagement in determining the scope and support for national programs, prioritizing critical services without compromising the overall care quality, and securing clarity and guarantees in new funding approaches, including the bilateral funding MoUs with the United States Government
Decentralizing Diagnosis of Drug Resistance Testing: Insights for Effective Implementation
Timely and accurate diagnosis is essential for the effective management of drug-resistant tuberculosis (TB). The WHO recommends low-complexity automated nucleic acid amplification tests (e.g. Xpert MTB/RIF Ultra, Truenat MTB and MTB-RIF Dx) for the initial detection of tuberculosis and rifampicin resistance as well as for the detection of resistance to isoniazid and second-line drugs (Xpert MTB/XDR). This session, organised in collaboration with Cepheid, highlighted Ethiopia’s implementation experience in expanding access to Xpert MTB/XDR for the early identification of pre-XDR/XDR patients. Dr Endale Mengesha, Senior TB Laboratory Diagnostic Services Advisor, emphasized that deploying Xpert MTB/XDR aligned with national priorities to provide all individuals at risk of drug-resistant infection with drug susceptibility testing. The deployment leveraged existing high-volume GeneXpert facilities to optimize infrastructure use. Site selection focused on locations with elevated rates of rifampicin resistance and high patient volumes. Furthermore, relevant policies and guidelines were revised to ensure Xpert MTB/XDR was the primary method for testing of susceptibility to isoniazid and second line drugs. Simultaneously, staff training, resource quantification and forecasting, enhancement of data management systems, and enrollment of sites into external quality assurance programs was done. By the end of the roll-out, the Xpert MTB/XDR assay had been deployed to 122 sites resulting in >98% of patients with rifampicin resistance being tested. However, challenges related to electricity and supply chain challenges (including consumables), data management, and staffing must be addressed to fully realize the advantages of rapid molecular testing for drug-resistant TB.
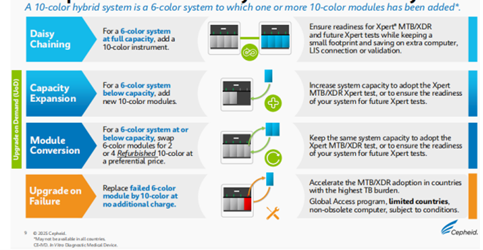
Pathways from 6-color to 10-color systems
Rapid diagnostics using low-complexity NAAT is required in closing the gap in early TB and drug resistance detection.
GIS Mapping of Laboratory Systems: Launch of the Framework
The African Society for Laboratory Medicine (ASLM) and Africa Centres for Disease Control and Prevention (Africa CDC) launched the Framework for GIS Mapping of Laboratory Systems and Network Capacities in Africa. The framework aims to optimize diagnostic networks and strengthen health systems through standardized mapping practices, emphasizing country ownership and sustainable, nationally-led programs.
This framework launch and dissemination session further highlighted r the crucial need valid lab network mapping data to guide national planning, surveillance, national sample transport network logistics, and diagnostic network optimization. The launch also covered proposed sampling strategies for countries that may not have adequate resources to map all laboratories yet require mapping data for informed decision making.
The final segment of this session focused on experience sharing from Cameroon and Tunisia. The two countries highlighted practical application of the framework, in their hybrid training and data collection, data use in the update of national policy and guideline documents and the scale up of mapping to lower-level laboratories.
